
Keepinmindthatintheseexamplesweareneglectingthepresenceofanynon-bondingelectrons(also known as “lone pairs”). This particular arrangement results in what is called acarbonateion. Therefore, in this molecule there are two oxygen atoms with a formal chargeof+2andonecarbonatomwithaformalchargeof0. Therefore, we can calculate the formal charges as follows:įormal Charge on Oxygen = 6 – (3 + 1) = 2įormal Charge on Carbon = 4 – (2 + 2) = 0 In this molecule, each oxygenatom has sixvalenceelectrons (three lone pairs and one bond), whilecarbonhasfourvalenceelectrons(two bondsand two lone pairs). For example, consider this Lewis dot structure for carbon dioxide: O=C=O. The resulting number is the formal charge on that atom. įormal charge is calculated by subtracting the number of valence electrons on an atom from the total number of electrons surrounding that atom in a Lewis dot structure. For example, carbon has four valence electrons, so its Lewis dot structure would look like this: C. The number of dots equals the number of valence electrons for that particular atom. In a Lewis dot structure, each dot represents one valence electron. The concept of formal charge is based on Lewis dot structures, which are two-dimensional representations of molecules and ions that show how valence electrons are arranged around atoms. 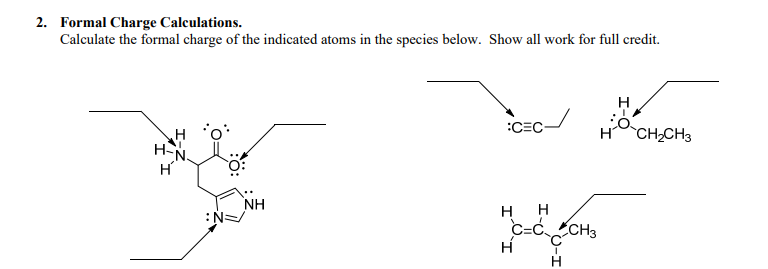 
It also provides insight into possible electron-sharing arrangements (resonance structures) in molecules and how those resonance structures contribute to the overall stability of the molecule. Formal charge is important because it allows chemists to predict the predominant structure or ionic character of a molecule or polyatomic ion. In chemistry, formal charge is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. This will give you your final answer for formal charge. Finally, you need to subtract the total number of lone pairs from the total number of bonding electrons. This will give you the number of bonding electrons in the molecule. Once you have determined the total number of valence electrons, you need to subtract the number of non-bonding electrons from this total. This can be done by looking at the periodic table or using an online tool. The first thing that you need to do is determine the total number of valence electrons in the molecule. 
This will ensure that you get the correct answer every time. Since sulfur has six valence electrons, we conclude that two electrons are not involved in the bonding, i.e., that there is a lone pair.When it comes to calculating formal charge, there is a simple formula that you can follow. In the sulfite ion, SO 3 2– for example, the oxidation number of sulfur is +4, suggesting that only four sulfur electrons are involved in the bonding. Oxidation numbers can sometimes also be useful in writing Lewis structures, particularly for oxyanions. This type of reaction can be recognized because it involves a change in oxidation number of at least one element. Oxidation numbers are mainly used by chemists to identify and handle a type of chemical reaction called a redox reaction, or an oxidation-reduction reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
